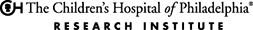

An international scientific team discovered a gene associated with a high risk of severe, recurring childhood asthma, a leading cause of hospitalization in young children. Identifying a risk susceptibility gene linked to this phenotype may lead to more effective, targeted treatments for this type of childhood asthma, said co-lead author of the study, Hakon Hakonarson, MD, PhD, director of the CHOP Center for Applied Genomics (CAG).
The study team, which included collaborators from five countries, performed a genome-wide association study on DNA from Danish national health registries and the Danish National Screening Biobank. In the discovery phase of their study, they compared genomes from 1,173 children aged 2 to 6 years from the Copenhagen Prospective Study on Asthma in Childhood with genomes from 2,522 adult and pediatric control subjects without asthma. The study team replicated their findings using data from other children of both European and non-European ancestry.
The researchers identified a novel gene, CDHR3, which is particularly active in epithelial cells lining the surfaces of airways.
“Abnormalities in the epithelial cells may increase a patient’s risk to environmental triggers by exaggerating immune responses and making the airway overreact,” said Dr. Hakonarson, a pediatric pulmonologist. “Because the CDHR3 gene is related to a family of proteins involved in cell adhesion and cell-to-cell interaction, it is plausible that variations in this gene may disrupt normal functioning in these airway cells and make a child vulnerable to asthma.”
The findings from the study, published in Nature Genetics, were consistent with previous investigations by CAG, suggesting that other genes linked to childhood asthma play a role in oversensitive immune reactions. Further research is needed to better understand how the CDHR3 gene may function in asthma.

At birth, infants move from a sterile environment to one full of microorganisms. Researchers at Children’s Hospital used mouse models to show that changes in microbial colonization of the gastrointestinal tract play a vital role in this transition. Ultimately, this research may lead to potential treatments to restore critically ill newborns’ resistance to common pathogens.
“Babies who are born preterm, in addition to them having less ability to fight infection, are more likely to get infected,” said Hitesh Deshmukh, MD, PhD, of CHOP’s Division of Neonatology.
Dr. Deshmukh and his colleagues found that regulation of postnatal granulocytosis — a burst in production of infection-fighting white blood cells that infants experience within 12 to 24 hours after birth — likely lies within the gut microbiome. Microbial colonization of the gut starts upon an infant’s arrival into the world, initiating this immune response. The investigators showed that antibiotics hampered the natural buildup of granulocytes in mice models.
“While some antibiotics may affect the bone marrow directly, we believe these effects are due to the influence of antibiotics on bacteria in the gut,” he said. “If you were to replace some of those microbes, you might restore the resistance of the newborn to the infection.”
In a study published in Nature Medicine, his research team proved this by taking normal intestinal microbiota from mice that were not exposed to antibiotics and transferring them to mice that had received antibiotics. In adult humans, this procedure is called a fecal transplant.
“Fecal transplant could be one of the ways in which you could make sure that babies have more resistance or more power to fight infection,” said the study’s senior author G. Scott Worthen, MD, a professor of pediatrics in the Division of Neonatology at the Perelman School of Medicine at the University of Pennsylvania. “One of the things you could do with this mixture is to use it to rapidly reconstitute an infant after they’ve finished a course of antibiotics.”
Dr. Deshmukh’s future research will focus on identifying bacterial components that could generate a new microbial community for preterm infants who needed antibiotics and subsequently preserve or restore their resistance to infection.
Neonatologists commonly use noninvasive nasal ventilation instead of mechanical ventilation via a breathing tube, in hopes of avoiding bronchopulmonary dysplasia (BPD). A study led by CHOP investigator Haresh Kirpalani, BM, showed that current noninvasive techniques for respiratory support are less effective than assumed in reducing the incidence of severe lung injury in very premature infants.
Frequently a byproduct of intubation, BPD — scarring and inflammation of the lungs — is a leading cause of death or neurological injury in infants with extremely low birth weights.
Dr. Kirpalani and his team compared two common forms of noninvasive ventilation used in extremely-low-birth-weight premature infants. Both techniques make breathing easier for the infant by stopping the lungs from collapsing, which over time causes lung inflammation and injury.
The current standard of care, nasal continuous positive airway pressure (CPAP), delivers slightly pressurized air throughout the breathing cycle. In contrast, nasal intermittent positive-pressure ventilation (IPPV), which has become widespread, provides an additional spike of positive pressure when the infant inhales. While more complicated, the hope had been that IPPV was more effective than standard CPAP.
With this study, published in the New England Journal of Medicine, Dr. Kirpalani and his team found no significant difference in the primary outcome of either death or survival with BPD at 36 weeks. They also found no significant difference in rates of other neonatal complications.
Most existing vaccines use inactivated viruses or similar particles to stimulate the body's immune system to release infection-fighting antibodies; however, rapidly evolving infectious agents such as HIV can evade traditional vaccine candidates. As a result, vaccine experts have been working to develop next generation vaccines that elicit broadly neutralizing antibodies to strike against hidden vulnerable structures within quick-changing viruses.
An advanced approach to vaccine design could be useful against highly variable viruses that have been difficult to stop using traditional strategies. Philip R. Johnson, MD, chief scientific officer and director of the Research Institute, was among the co-authors of a study published in Nature that offers proof-of-principle that a new software application, “Fold from Loops,” can custom-design artificial proteins as vaccine components.
The new strategy uses sophisticated techniques to imitate an epitope — a structure specific to each type of invading virus that is recognized by the immune system. “Fold from Loops” designs flexible protein scaffolds to hold the epitope that induces the immune system to produce protective antibodies.
In the Nature study, the research team induced potent antibodies in non-human primates against respiratory syncytial virus (RSV), a serious childhood infection. The successful experiment demonstrates that this approach is feasible for developing a first RSV vaccine in humans, as well as for accelerating the creation of new vaccines against difficult-to-treat diseases such as HIV, influenza, and hepatitis C.
“Bringing these new types of vaccines into clinical use will take years of work, but this study represents an important first step along the way,” Dr. Johnson said.

Primary mitochondrial diseases directly interfere with the function of the respiratory chain (RC) — the highly conserved sequence of chemical reactions within mitochondria that generate energy from oxygen and nutrients. RC malfunction in mitochondrial disease may cause symptoms such as seizures, strokes, blindness, heart disease, progressive muscle weakness, and vulnerability to infections. No cure exists, and most current treatments for RC diseases are largely ineffective.
CHOP investigators identified a master network of signaling molecules that acts like a “fuse box” to regulate the cellular effects of defective energy flow in mitochondrial RC diseases — a diverse set of difficult-to-treat genetic-based energy disorders. Using that knowledge, they showed that a form of vitamin B3 called nicotinic acid partially restores normal functioning in cells taken from patients with mitochondrial disease.
Marni J. Falk, MD, led the study published in the journal PLOS ONE suggesting that the regulatory signaling network may offer a common avenue to target in developing effective, personalized treatments for many mitochondrial energy disorders.
The investigators analyzed cellular responses in human skeletal muscle and skin cell lines, finding that RC disease disrupted crucial biological pathways controlled by a handful of master signaling factors. All of those factors are integral components of cellular signaling networks that sense nutrient availability and regulate growth.
Building on her team’s previous studies, Dr. Falk and colleagues added nicotinic acid to a cell line grown from the skin of a patient with the mitochondrial disease known as Leigh syndrome that causes strokes in young children.
The results were exciting. The nicotinic acid normalized signaling activity across an integrated signaling network, and it also improved overall cellular respiration — the cells’ ability to use oxygen.

Parents and primary care physicians should consider that children with traumatic brain injury (TBI) may need ongoing support to address difficulties with memory, problem solving, and verbal communication when transitioning from inpatient rehabilitation to home and school, according to a study conducted by researchers at Children’s Hospital.
The study published in the Journal of Pediatrics included data from nearly 14,000 children aged 7 to 18 years with trauma-related injuries who completed inpatient rehabilitation during the period from 2002 to 2011.
The investigators, led by Mark Zonfrillo, MD, MSCE, analyzed the children’s cognitive deficits before and after admission to inpatient rehabilitation. Children with TBI had more cognitive disability when entering inpatient rehab than other children with serious injuries, which included spinal cord injury, multiple fractures, amputations, and burns. While all injury groups showed improvements upon discharge, children with TBI had more residual cognitive disability.
These children may continue to improve after discharge, so this transition is a critical point in their care, said Dr. Zonfrillo, who is an investigator at CHOP’s Center for Injury Research and Prevention and associate director of research in the Division of Emergency Medicine.
“The goal of this line of research at CHOP is to maximize access to trauma systems and to optimize acute and rehabilitation care for young patients with serious injuries,” Dr. Zonfrillo said. “If we have a complete picture of the scale of pediatric traumatic injuries and related long-term needs, as well as the differences between injury patterns and various outcomes, we can predict the type and level of support necessary upon discharge from inpatient rehabilitation.”
Hematologists have long sought to reactivate fetal hemoglobin as a treatment for children and adults with sickle cell disease (SCD), a painful, sometimes life-threatening genetic disorder that deforms red blood cells and disrupts normal circulation.
Shortly after birth, regulatory elements in DNA shift the body from producing the fetal form of hemoglobin to producing the adult form instead. But when patients with SCD undergo this transition, their inherited gene mutation distorts adult hemoglobin, forcing red blood cells to assume a sickle shape.
CHOP hematology researcher Jeremy W. Rupon, MD, PhD, who worked in collaboration with a former postdoctoral fellow, Wulan Deng, PhD, in the laboratory of Gerd Blobel, MD, PhD, reprogrammed gene expression to reverse the biological switch. This caused cells to resume producing fetal hemoglobin, which is not affected by the SCD mutation, and produces normally shaped red blood cells.
“Our study shows the power of a technique called forced chromatin looping in reprogramming gene expression in blood-forming cells,” Dr. Rupon said. “If we can translate this approach to humans, we may enable new treatment options for patients.”
Dr. Blobel’s team demonstrated in previous work that chromatin looping, a tightly regulated interaction between widely separated DNA sequences, drives gene transcription — the conversion of DNA code into RNA messages to carry out biological processes.
In the current study, the investigators custom-designed a genetically engineered zinc finger protein that latched onto a specific DNA site carrying the code for fetal hemoglobin. They attached the zinc finger to another protein that forced a chromatin loop to form. The loop then activated gene expression that produced embryonic hemoglobin in blood-forming cells from adult mice. The team obtained similar results in human adult red blood cells, forcing the cells to produce fetal hemoglobin.
Retinopathy of prematurity (ROP) is a disorder of the blood vessels of the retina, which are not completely developed until a baby reaches full term, and its incidence has been rising.
“It is therefore important to research the association between this neonatal complication and adverse long-term child development, ” said Barbara Schmidt, MD, MSc, an attending neonatologist at Children’s Hospital and also a professor of pediatrics and Kristine Sandberg Knisely Chair in Neonatology, Perelman School of Medicine at the University of Pennsylvania.
Dr. Schmidt led a study that showed infants with severe ROP diagnosed and treated under modern protocols remain at risk of nonvisual disabilities, even if blindness can be averted in most children. This exploratory analysis reported in the Journal of the American Medical Association used data from a cohort of very low-birth-weight infants involved in the Caffeine for Apnea of Prematurity trial.
The study found that motor impairment, cognitive impairment, and severe hearing loss were three to four times more common in children with severe ROP than those without severe ROP. Yet, it remains unclear why there is an association between the development of severe ROP and the presence of nonvisual disabilities.
“We can only speculate,” Dr. Schmidt said. “The retina has been called a ‘window to the brain;’ hence, severe damage to the developing retina in a very immature baby may also indicate damage to the developing brain.”
These findings remind clinicians and parents that while blindness often can be prevented by timely retinal therapy in the neonatal intensive care unit, severe ROP remains a predictor of functional disability. It reinforces the need for long-term visual and developmental follow-up for infants who are diagnosed with severe ROP.

Obstructive sleep apnea syndrome, or OSAS, is a condition of interrupted breathing caused by a narrowing in the throat or upper airway, resulting from enlarged tonsils and adenoids, obesity, or other medical problems. The primary treatment for OSAS in children is adenotonsillectomy, the surgical removal of the adenoids and tonsils. More than half a million U.S. children undergo the surgery each year.
Sleep experts conducted the first multicenter clinical trial of OSAS in children and found that those who underwent adenotonsillectomy had notable improvements in behavior, quality of life, and other symptoms compared to those treated with “watchful waiting” and supportive care. However, the researchers found no difference between treatment groups in attention and executive functioning, as measured by formal neuropsychological tests.
“This was the first randomized clinical trial of surgery for obstructive sleep apnea in children,” said first author Carole L. Marcus, MBBCh, who directs the Sleep Center at Children’s Hospital. “Some previous, smaller studies had found this condition associated with cognitive and behavioral problems, including ADHD, so it was important to do a controlled trial to evaluate the benefits of surgery, which is a common treatment.”
The findings are the result of the Childhood Adenotonsillectomy Trial (CHAT) that involved 464 children, aged 5 to 9 years, at seven academic sleep centers. The CHAT researchers published their findings in the New England Journal of Medicine, in conjunction with a presentation at the American Thoracic Society annual meeting.
Overall, both surgery and watchful waiting were safe in this clinical trial, the researchers noted. Watchful waiting was a reasonable option for parents of children with less severe OSAS who opt not to have surgery, Dr. Marcus said, but clinicians should carefully monitor these children to ensure their condition does not worsen.
“Sleep medicine is a very new field, with many unanswered questions,” Dr. Marcus said. “For instance, we go to sleep each night, yet we don’t even understand the true purpose of sleep. But this study is a great first step in finding some of the answers.”

The good news is that doctors are able to cure nearly 80 percent of cancer patients, but that does not mean that those pediatric patients who survive cancer stop fighting once treatment ends.
Children’s Hospital’s Cancer Survivorship Program “helps patients and families navigate life after cancer, including both the physical and emotional issues they may have.” The program, which is led by Jill P. Ginsberg, MD, includes research into the neurobehavioral outcomes of acute lymphoblastic leukemia survivors and key adverse events following childhood cancer.
A research initiative led by Dr. Ginsberg and Thomas F. Kolon, MD, involving prepubescent boys who have been diagnosed with cancer, was the subject of media attention, with articles appearing in Nature Medicine and The Wall Street Journal.
An estimated 35 percent of prepubescent boys are at risk of sterility following cancer treatment, Dr. Ginsberg said, and because prepubescent boys cannot produce sperm, CHOP’s testicular tissue cryopreservation program can offer families hope that their tissue could one day be used to produce viable sperm. Only boys at the highest risk for infertility are approached to take part in the program because the procedure remains experimental.
Based on pioneering work by the University of Pennsylvania’s Ralph L. Brinster, PhD, the procedure involves the removal of a small amount of testicular tissue containing spermatogonial stem cells. Scientists suggest that one day these cells could either be transplanted back into the boys to grow spermatogonial tissue following cancer treatment or be used to grow sperm in vitro. Though researchers have yet to produce sperm using human spermatogonial tissue, work by Dr. Brinster’s lab has shown that the approach works in a mouse model.
A $125,000 St. Baldrick’s Foundation grant awarded in August 2013 will allow the Testicular Cryopreservation Consortium to expand beyond CHOP to include Seattle Children’s Hospital and Memorial Sloan-Kettering Cancer Center.
“We are hopeful that advances in the laboratory will make it possible for these boys to achieve fertility when they are ready to start a family,” said Dr. Ginsberg after the award was announced. “This work could not have been accomplished without the support of St. Baldrick’s Foundation.”
In addition to its groundbreaking program for prepubescent boys, CHOP is one of few, if not the only, institutions in the country that invite all postpubescent boys set to undergo cancer treatment to participate in a sperm-banking program. About 85 percent of boys agree to bank.
It is “one of the programs here at CHOP that I’m most proud of,” Dr. Ginsberg said.